
Fluorinated Carbon Synthesis. Professor Jong-Beom Baek’s UNIST team advances battery tech. They’ve devised a novel approach to safely produce fluorinated carbon materials (FCMs) by utilizing polytetrafluoroethylene (PTFE) and graphite.
Fluorinated carbon materials (FCMs) are stable due to strong C-F bonds. However, traditional methods for fluorination rely on highly toxic reagents like hydrofluoric acid (HF), making them impractical for widespread use.
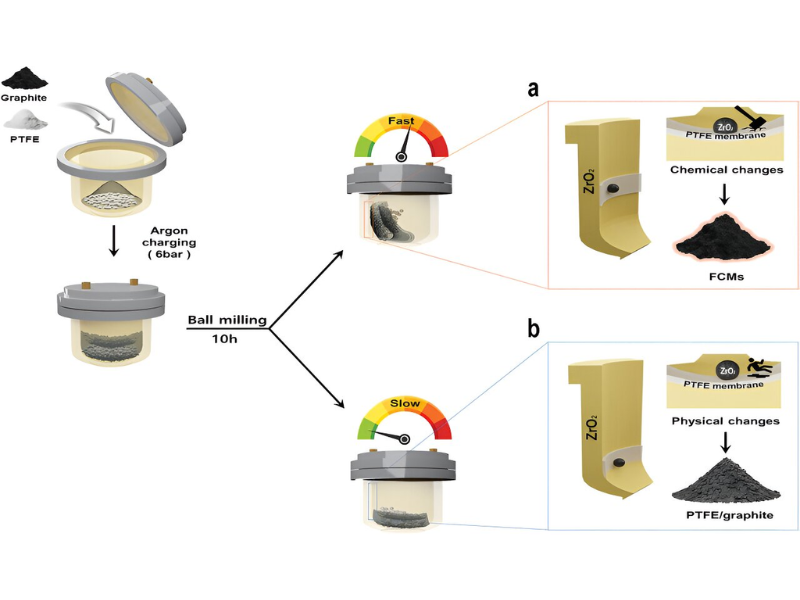
In this study, the research team introduced an accessible and relatively safe approach for scalable FCM synthesis. They achieved this by employing mechanochemical depolymerization of PTFE, a common compound found in everyday items, and the fragmentation of graphite. Using ball-milling techniques that induce both mechanical and chemical reactions, they successfully generated FCMs with markedly improved performance compared to graphite.
Conventional carbon fluoride production methods
Conventional carbon fluoride production methods involve hazardous compounds like fluorine gas or HF, which raise safety concerns and lead to increased manufacturing costs due to stringent safety measures. To tackle these challenges, Professor Baek’s team developed a solid-phase fluorination method utilizing PTFE—an inert polymer known for its stability in atmospheric conditions and safety when ingested.
Experiments showed that excessive energy caused PTFE molecular chain breakage and radical formation. This triggered a reaction forming carbon fluoride complexes on graphite particles.
The resulting FCMs exhibited significantly improved storage capacity and electrochemical stability in comparison to conventional graphite anodes. At a low charging rate of 50 mA/g, the FCMs displayed storage capacities 2.5 times higher (951.6 mAh/g) than graphite. Moreover, at a high charging rate of 10,000 mA/g, their storage capacity was tenfold greater (329 mAh/g).Even after 1,000+ charge/discharge cycles at 2,000 mA/g, FCMs retained 76.6% of capacity, compared to graphite’s 43.8%.
Jang, a UNIST researcher, highlights the broader potential of solid-phase reactions. Professor Baek emphasizes solid-phase reactions’ potential to discover novel materials.
The study’s findings have been published in Advanced Functional Materials.
Read the original on Phys.
Read more: Quantum Device Slows Simulated Chemical Reaction by 100 Billion Times.
Comments
One response to “Direct Synthesis of Fluorinated Carbon Materials through Mechanochemical Reaction”