
A new study found that stress experienced during pregnancy can rewire a baby’s brain, increasing the risk of anxiety. Negative conditions before birth alter the development of neurons in the hippocampus, making the child more sensitive to threats later in life.
According to the World Health Organization (WHO), anxiety disorders are the most prevalent mental health condition worldwide. Although reacting to danger is a natural survival mechanism, overreacting to perceived threats is harmful. This raises the question: are these heightened reactions inherent from birth or acquired through experience?
Prenatal Stress May Rewire the Brain, Increasing Risk of Anxiety Later in Life
Researchers at Weill Cornell Medicine (WCM) launched a new study to examine whether exposure to adverse conditions during development—such as inflammation and infection—leads to anxiety later in life.
“Our findings show that prenatal adversity causes lasting changes in the neurons of the ventral dentate gyrus (vDG), connecting the gestational environment to anxiety-like behavior,” said Professor Miklos Toth, MD, PhD, co-corresponding author and Professor of Pharmacology. “This process could help explain why some individuals with innate anxiety exhibit ongoing stress sensitivity and avoidance.”
The ventral dentate gyrus (vDG), a part of the hippocampus, actively regulates emotional processing and stress responses, linking it closely to anxiety. To study this, researchers used a mouse model to replicate an adverse prenatal environment, specifically maternal immune activation, which simulates infection or inflammation during pregnancy, and observed its impact on the vDG cells.
Prenatal Stress May Rewire the Brain, Increasing Risk of Anxiety Later in Life
They used a range of techniques—conducting behavioral tests, recording neuronal firing and communication through electrophysiology, analyzing DNA methylation to track gene regulation changes, sequencing RNA to identify activated or suppressed genes in specific neurons, and applying fiber photometry to monitor real-time neural activity in both safe and threatening situations.
Mouse offspring exposed to stressful or adverse conditions during pregnancy exhibited increased anxiety-like behaviors. In these mice, the researchers found that ventral dentate granule cells (vDGCs) experienced decreased inhibition, resulting in heightened excitability. The stressful prenatal environment altered gene expression in the offspring’s neurons by modifying chemical markers on their DNA (DNA methylation). These changes primarily impacted genes involved in neuronal connectivity and communication.
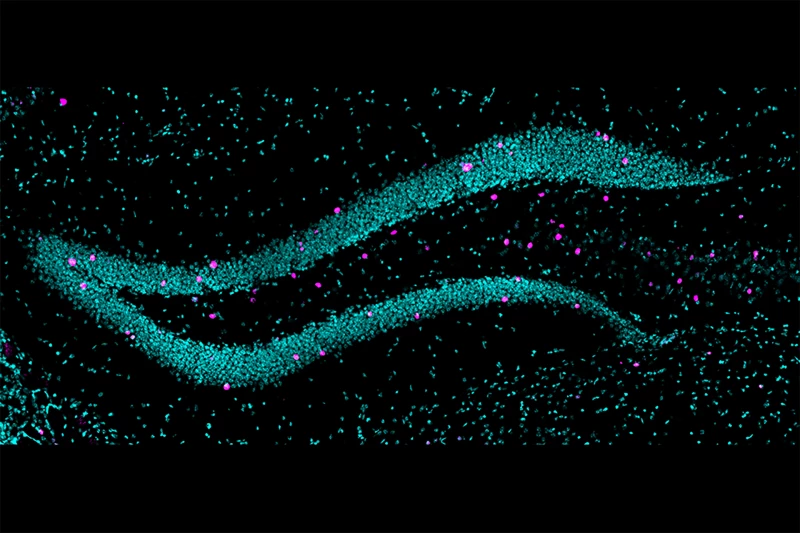
Weill Cornell Medicine/Nicole Politowska
When the mice were placed in stressful or anxiety-inducing situations, the brain cells with these changes became the most active. In these cells, many genes related to neural connections were functioning differently than usual. Real-time observations of the ventral dentate granule cells (vDGCs) revealed that mice exposed to adversity showed heightened activity during shifts from safe to threatening environments, indicating an increased perception of threat.
“Overall, these epigenetic modifications cause specific neurons in the vDG to react differently in adulthood when encountering unsafe environments,” explained Kristen Pleil, PhD, Associate Professor of Pharmacology at WCM and the study’s co-corresponding author. “These neurons become overactive, leading the mice to interpret their surroundings as more dangerous than they really are.”
Study Limitations Acknowledge Need for Research, But Strengthen Prenatal Stress–Anxiety Link
The study has some limitations. Because the researchers conducted the study on mice, the findings may not translate directly to humans. The effects varied among neurons, with only about 10% to 30% of cells showing significant methylation changes. Additionally, the study did not definitively establish which methylation changes led to specific transcriptomic alterations, a question that requires further investigation. The research focused on the ventral dentate gyrus (vDG), though other parts of the hippocampus might also play a role.
Despite these limitations, the study supports the notion that maternal inflammation or stress during pregnancy can increase the risk of anxiety disorders in offspring later in life. The researchers suggest that their work offers one of the clearest mechanistic connections so far between the prenatal environment, epigenetic modifications, and psychiatric conditions.
“A mouse’s ventral dentate gyrus (vDG) contains nearly 400,000 cells, yet only a few thousand are affected during pregnancy,” Toth explained. “Our next goal is to uncover why these specific cells undergo epigenetic programming.”
Read the original article on: New Atlas
Read more: Pregnancy-Simulating Robot Debuts in China


