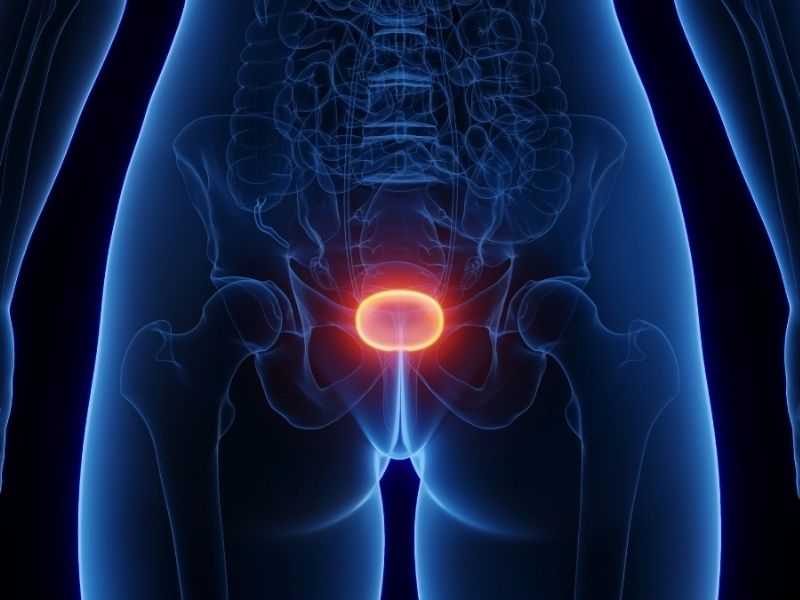
Urology departments in England and Wales have observed a rise in hospital admissions for bladder inflammation among 16- to 24-year-olds linked to ketamine use.
This trend seems to reflect a broader increase in ketamine consumption, with the number of young people and adults seeking treatment for ketamine abuse last year rising sharply compared to previous years.
Ketamine’s Impact on the Bladder
Ketamine misuse can significantly affect the bladder, leading to symptoms such as frequent urination, nighttime urination, sudden urges, leakage, inflammation, bladder or lower back pain, and blood in the urine.
These effects can be severe, disrupt daily life, and in some cases, may cause lasting damage.

Ketamine was originally approved in 1970 for use as a human anesthetic. More recently, research has indicated that low doses of ketamine may have antidepressant properties.
However, recreational use of ketamine has been increasing.
As a dissociative drug, it can make users feel disconnected from themselves and their environment. It also produces hallucinogenic, stimulant, and pain-relieving effects, typically lasting one to two hours.
People usually take ketamine by snorting or smoking the powder, injecting the liquid form, or mixing it into drinks. Snorting generally leads to stronger effects and more pronounced symptoms than ingesting it orally.
People who use ketamine can build up a tolerance rapidly, requiring larger doses to achieve the same effects. This likely happens because the body and brain adapt, becoming more efficient at metabolizing the drug.
Bladder Injury
Regular use of high doses of ketamine can lead to serious harm to the bladder, urinary system, and kidneys. In extreme cases, surgical removal of the bladder may be necessary.
The earliest documented cases of ketamine-related bladder damage were reported in Canada in 2007, involving nine recreational users who experienced severe bladder issues and blood in their urine. A later, larger study in Hong Kong observed similar problems in 59 individuals who had used ketamine for over three months.
Like all drugs, ketamine is processed by the body, broken down, and eventually eliminated through the urine.
Understanding Ketamine’s Effects on Bladder Health
When ketamine is metabolized, it produces substances that can seriously harm the bladder. Prolonged contact of these by-products with the urinary tract irritates and damages the tissues.
The bladder is affected first because it stores urine the longest. Over time, the ureters (the tubes connecting the kidneys to the bladder) and the kidneys may also become damaged.
Chronic use can cause the bladder to shrink and stiffen, leading to severe urinary symptoms. The ureters may narrow and twist, sometimes taking on a “walking stick” shape, which can cause urine to back up into the kidneys, a condition known as hydronephrosis.
Cell Damage and Overactive Bladder from Ketamine
Ketamine also raises oxidative stress, harming cells and causing bladder cells to die. This disrupts the bladder’s protective lining, making it more permeable and highly sensitive.
These changes can lead to an overactive and painful bladder, often causing intense urges to urinate and urinary incontinence.
Bladder damage from ketamine develops gradually in stages.
In the first stage, the bladder becomes inflamed. This damage is often reversible by stopping ketamine and using medications such as anti-inflammatories, pain relievers, or prescription drugs that reduce urgency and support healing of the bladder lining.
In the second stage, the bladder may shrink or become stiff. Treatment is similar to the first stage, but may also include a bladder instillation—a procedure in which a catheter delivers medication directly into the bladder. This medication coats the inner lining, helping to restore its protective layer and reduce inflammation.
Botulinum toxin injections can also be used to relax the bladder and reduce pain and urgency. However, stopping ketamine use is crucial to prevent further damage.
In the final stage, permanent harm can occur to both the bladder and kidneys. If the kidneys become affected over time, this may lead to kidney failure. Treatments such as dialysis—which filters waste and excess fluid from the blood—or even surgery may be needed to restore kidney and urinary function.
Although ketamine has been classified as a Class B drug since 2014, it remains cheap and easy to obtain, costing as little as £3 per gram in some areas of the UK. Increasing awareness of ketamine’s health risks is vital to help prevent these serious complications.
Read the original article on: Sciencealert
Read more: New Research Reveals a Way to Reactivate T Cells That Destroy Cancer