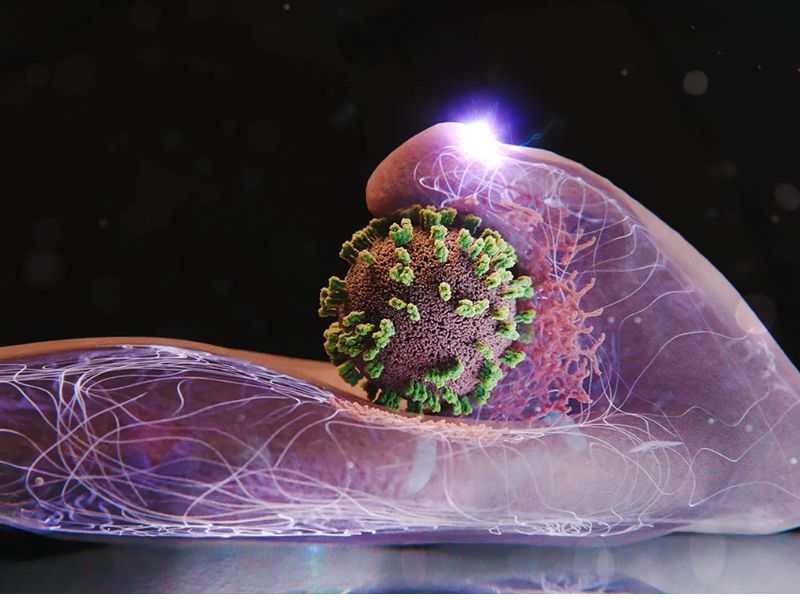
A research team from Switzerland and Japan has closely examined how the virus behaves. Using a self-developed microscopy technique, the scientists can focus on the outer surface of human cells in a Petri dish, allowing them to observe in real time the precise moment an influenza virus enters a living cell.
Led by Yohei Yamauchi, Professor of Molecular Medicine at ETH Zurich, the team made a surprising discovery. Rather than remaining passive as the influenza virus nears, the cells seem to actively try to capture it. “The infection of our body’s cells is like a dance between virus and cell,” explains Yamauchi.
How Viruses Exploit Essential Pathways
Although cells gain no benefit from infection, the interaction appears active because the virus takes advantage of a normal cellular uptake system that the cells rely on. This system typically transports vital substances like hormones, cholesterol, and iron into the cell.
To start the infection, an influenza virus binds to specific molecules on the cell surface. It “surfs” along the membrane, attaching to one molecule after another until it reaches an area dense with these receptors. A cluster of receptors provides the most efficient entry point.
Once the receptors detect the virus, the membrane begins to form a small indentation at that site. A structural protein called clathrin shapes and reinforces this pocket. As the pocket deepens, it envelops the virus and forms a vesicle. The cell then pulls the vesicle inward, where the clathrin coat disassembles and releases the virus inside.
Why Traditional Imaging Falls Short in Capturing Viral Entry
Earlier efforts to examine this critical stage of infection depended on techniques such as electron microscopy, which destroy the cells to produce an image, capturing only isolated snapshots. Fluorescence microscopy, another frequently used method, allows live imaging but provides only low spatial resolution.
The new technique, named virus-view dual confocal and AFM (ViViD-AFM), combines atomic force microscopy (AFM) with fluorescence microscopy. This integrated method allows researchers to observe the detailed movements of the virus as it penetrates the cell.
Using this technique, the researchers showed that cells play an active role in multiple stages of viral entry. They recruit key clathrin proteins to the site where the virus is bound, and the membrane rises at that spot, seemingly attempting to capture the virus. These wave-like movements become stronger if the virus begins to move away from the cell surface.
Since ViViD-AFM enables real-time observation of infection, it provides a useful method for testing potential antiviral drugs directly in cell cultures. The researchers also suggest that this technique could be applied to studying other viruses or vaccines, allowing scientists to see how these particles interact with cells as it happens.
Read the original article on: Sciencedaily
Read more: An Aerial Microrobot Can Reach Speeds Similar To a Bumblebee’s