
Scientists in New Zealand have developed a minimally invasive technique that successfully restored movement in paralyzed rats. This breakthrough brings us closer to treating incurable spinal cord injuries that cause lasting motor loss in humans and animals.
Ultra-Thin Implant Delivers Targeted Spinal Cord Stimulation
The University of Auckland team created an ultra-thin implant that delivers precise electrical stimulation to a rat’s spinal cord injury.
Spinal cord injuries interrupt the brain’s communication with the body, largely because this nerve tissue doesn’t naturally regenerate well. To address this, the implant is designed to promote healing and help restore lost functions, explained Professor Darren Svirskis.
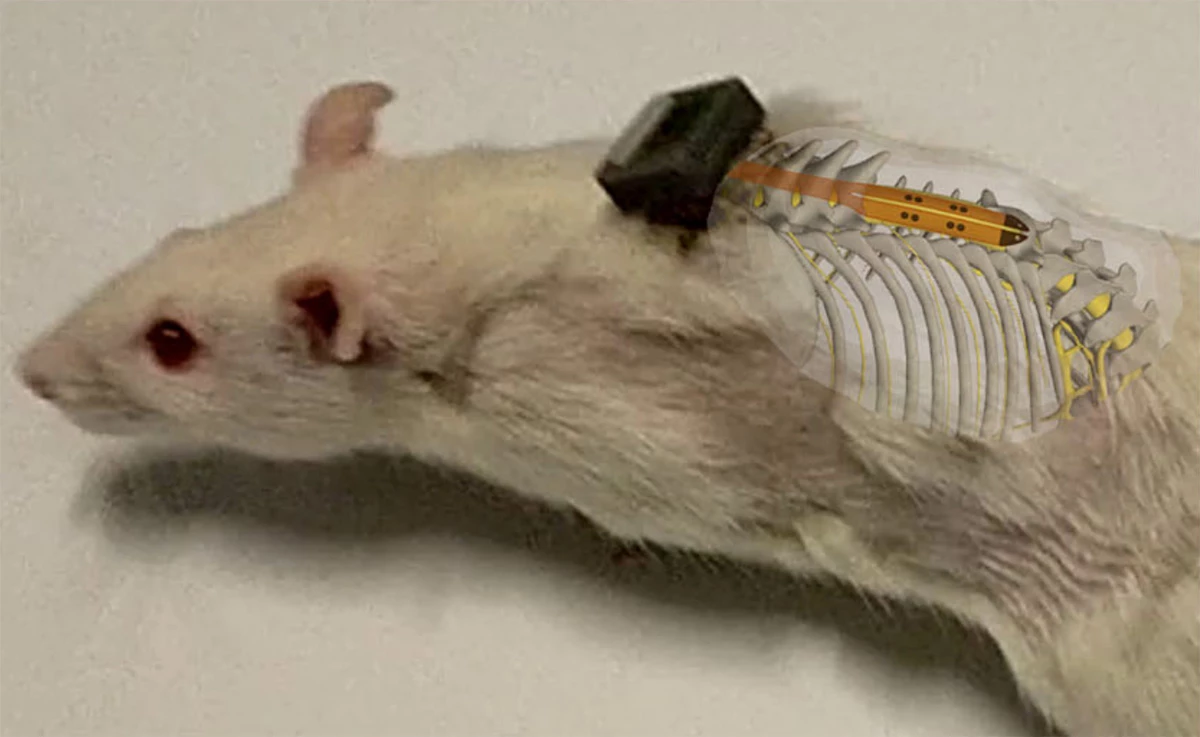
Image courtesy of the researchers
Custom biocompatible implants used low-frequency (2 Hz) electrical stimulation to promote nerve regeneration and new connections in damaged spinal cords.
Researchers treated one group of rats with moderate spinal injuries using daily electric field therapy for 1 hour over 7–11 days, then 5 days a week for 12 weeks. The control group was left to recover without any treatment.

Image courtesy of the researchers
The electric field (EF) treatment led to marked improvements compared to natural recovery. Rats that received the stimulation showed significantly better hind limb function after four weeks, with improved coordination, paw placement, and toe clearance. They also responded more quickly to mechanical touch, suggesting their sense of touch was returning. A video included with the team’s recent paper in Nature Communications shows the clear contrast between treated and untreated rats.
A 2012 Swiss study used chemical injections, electrical stimulation, and rehab on paralyzed rats. The new method is minimally invasive, aids movement and sensation, and avoids spinal damage.
Rats’ Natural Recovery Raises the Bar for Human Trials
It’s important to note that rats naturally have a higher ability to recover from spinal cord injuries than humans. This allowed researchers to compare natural healing with treatment more easily but requires more extensive research before applying it to humans.
The research team—which includes collaborators from Sweden’s Chalmers University of Technology—is now investigating how different levels of low-frequency EF treatment affect recovery. Their long-term goal is to develop a medical device to help people with spinal cord injuries regain lost function.
Read the original article on: New Atlas
Read more:Apple May Tap Anthropic Or OpenAI For Siri In a Major Strategy Shift



