Picture a future where tooth loss no longer requires costly implants, ill-fitting dentures, or artificial bridges. Instead, a single treatment could trigger the natural regrowth of a tooth, offering a permanent biological solution. What once sounded like science fiction is now moving toward reality, driven by groundbreaking research in Japan.

In September 2024, Kyoto University and Kitano Hospital began the first human trials of a drug that blocks USAG-1, a protein that prevents extra teeth. If successful, the treatment could reach the market by 2030. This article reviews the breakthrough and its potential impact on global dentistry.
In September 2024, Kyoto University and Kitano Hospital began human trials of a drug that blocks USAG-1, the protein that stops extra teeth from forming. If progress continues smoothly, the medication could become commercially available by 2030. This article explores how the innovation could reshape global dentistry.

The protein USAG-1 (Uterine Sensitization-Associated Gene-1) functions as a natural “brake” on tooth formation. USAG-1 suppresses extra tooth growth. About 1% of people have congenital tooth agenesis, while over 90% of Japanese over 75 experience tooth loss from decay, injury, or disease.
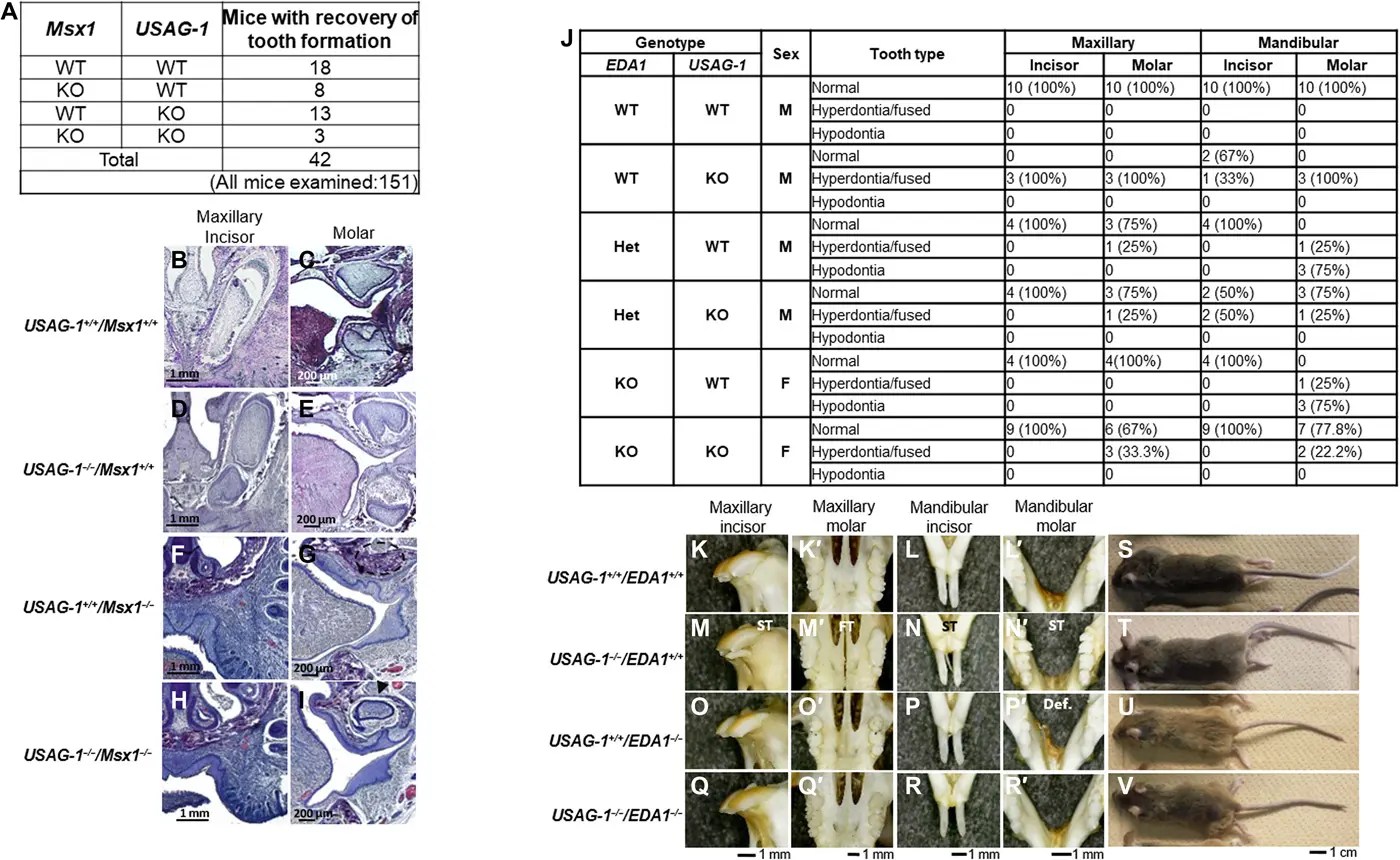
The experimental drug TRG-035, developed by Kyoto University spin-off Toregem Biopharma, uses monoclonal antibodies to block USAG-1 and reactivate BMP signaling, allowing dormant tooth buds to grow into complete teeth. In animal trials with mice and ferrets, a single intravenous dose successfully generated new teeth without major side effects.
Phase 1 Trials Underway at Kyoto University Hospital
Reports confirm that Phase 1 clinical trials began in September 2024 at Kyoto University Hospital. The trial involves 30 men aged 30–64 with at least one missing molar and will run until August 2025 to evaluate safety, dosage, and side effects. Participants receive intravenous doses, with progress monitored through imaging and clinical evaluations.
Updates from 2025 suggest encouraging results. By late 2024, the drug was tested in adults with acquired tooth loss, and Toregem Biopharma plans Phase 2 trials in 2025 for children aged 2–7 with oligodontia.Affecting about 0.1% of the population, this disorder can lead to nutritional and developmental challenges.
Dr. Takahashi’s Vision and Toregem’s Backing
Dr. Takahashi, who has spent nearly three decades studying tooth regeneration, stated: “We want to help people struggling with missing teeth. There is no permanent cure today, but hopes for tooth regrowth are strong.” Supported by over 100 collaborators, Toregem has funding from AMED and partners like WuXi Biologics. In 2023, Toregem raised 380 million yen to accelerate its research and development.
By 2025, developments remain promising. Early 2025 reports confirmed no major side effects in trials, and Toregem Biopharma presented findings at global events, highlighting potential benefits for older adults.
Still, challenges persist. Experts caution that animal trial results may not translate to humans, calling claims of a “third set of teeth” controversial. Concerns include ensuring correct tooth positioning to avoid misalignment and confirming long-term durability. Initial treatments may cost as much as implants (US$3,000–5,000 per tooth), though insurance could ease expenses.
Ethical and accessibility issues also play a role.Although early use targets rare conditions (1%), the long-term goal is treating common cases like cavities; in Brazil, where 15% of adults have tooth loss, this could cut public health costs.
TRG-035 as a Third Alternative to Dentures and Implants
If approved, TRG-035 could become a “third option” alongside dentures and implants. For children with agenesis, it would remove the need for frequently replaced dentures during growth. For adults and seniors, it could restore natural chewing, improving digestion and overall quality of life. Globally, the treatment has potential to help the estimated 5% of people living with partial tooth loss.
Meanwhile, complementary research is advancing. At King’s College London, scientists are testing Tideglusib to stimulate dentin regeneration in cavities using biodegradable sponges. Together, these innovations point toward a future of regenerative dentistry, where natural biology replaces synthetic solutions.
The outlook is credible and supported by reliable sources such as The Mainichi, New Atlas, and PubMed. With clinical trials ongoing and a projected launch around 2030, the USAG-1 inhibitor marks a major milestone. That said, patience is essential—scientific progress is deliberate to guarantee safety. Until then, good oral hygiene and regular dental care remain the best defense. Still, the day may come when regrown teeth are not the exception, but the standard in dentistry.
Read the original article on: Up Date Ordie
Read more: Waymo expands to Denver & Seattle