TU Wien (Vienna) has done well in making a new sort of material usable for chip technology with germanium and silicon. This allows faster, more efficient computers and new kinds of quantum devices.
Current chip technology is greatly based on silicon. Only in extremely special components, a small quantity of germanium is added. There are great reasons to utilize higher germanium contents later on: The compound semiconductor silicon-germanium has crucial advantages over today’s silicon technology regarding energy efficiency and achievable clock frequencies.
The main problem here is to dependably establish contacts between metal and semiconductors on a nanoscale. This is much more challenging with a high percentage of germanium than with silicon. The TU Wien team, together with study teams from Linz and Thun (Switzerland), has now revealed that this trouble can be fixed– with contacts made from crystalline aluminum of incredibly high quality and a sophisticated silicon germanium layer system. This allows different intriguing contact properties– especially for optoelectronic and quantum components.
The problem with oxygen
” Every semiconductor layer is immediately infected in conventional processes; this can not be stopped at the atomic level,” says Masiar Sistani from the Institute for Solid State Electronics at TU Wien. Firstly, oxygen atoms accumulate very rapidly on the surface of the materials, creating an oxide layer.
Nonetheless, this is not an issue with silicon: silicon always develops the same type of oxide. “With germanium, nevertheless, things are a lot more challenging,” explains Masiar Sistani. “In this case, there is an entire range of different oxides that can develop. That implies that different nanoelectronic devices can have extremely different surface compositions and consequently different electronic properties.”
Suppose you now intend to connect a metallic contact to these components. In that case, you have an issue: Even if you try hard to produce all these components precisely similarly, there are still unavoidably large differences, making the material complicated to handle for use in the semiconductor industry.
“Reproducibility is a big problem,” states Prof. Walter Weber, the head of the Institute for Solid State Electronics, TU Wien. “If you utilize germanium-rich silicon germanium, you can not make sure that the electronic component, after placing the contacts on it, will have the attributes you need.” Consequently, this material is just utilized to a limited extent in chip production.
That is a shame because silicon germanium would have definitive advantages: “The charge carrier concentration is much higher, especially positive charge carriers, the supposed ‘holes,’ can move much more efficiently in this material than in silicon. The material would consequently permit greater clock frequencies with increased energy efficiency than our current silicon chips,” says Lukas Wind, a doctoral student in Walter Weber’s study group.
The ‘perfect’ interface
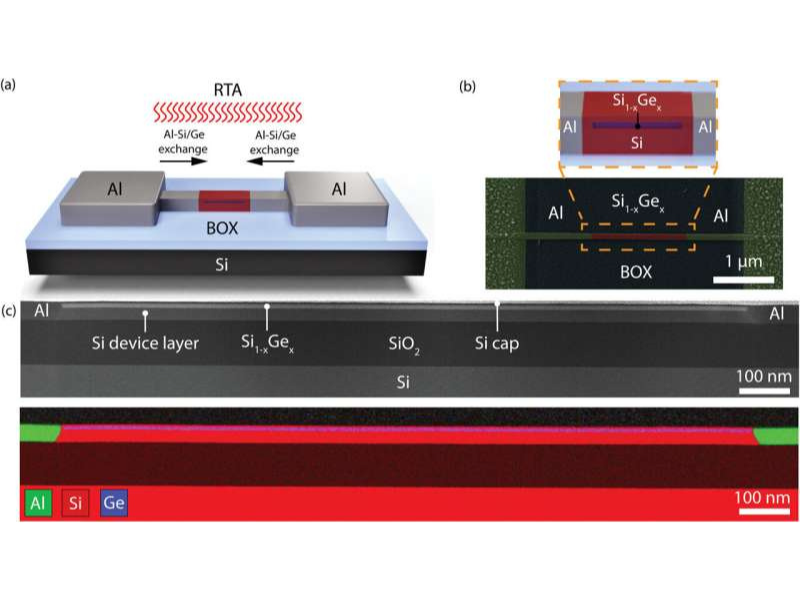
The study team has demonstrated how the issue can be fixed: They discovered a method to produce perfect interfaces between aluminum contacts and silicon germanium components on an atomic scale. In a primary step, a layer system is generated with a thin silicon layer and the actual material where the electronic components are to be made– the silicon-germanium.
By heating the structure in a controlled manner, a contact can currently be produced between the aluminum and the silicon: At around 500 degrees Celsius, unique diffusion occurs, and the atoms can leave their place and begin to migrate. Silicon and germanium atoms move into the aluminum contact relatively swiftly, and aluminum fills up the vacated space.
“The diffusion dynamics in the layer system used, therefore, produce an interface between aluminum and the silicon germanium with an extremely thin silicon layer in between,” clarifies Masiar Sistani. With this manufacturing process, oxygen atoms never ever have the opportunity to get to this atomically sharp and extremely pure interface.
“Our experiments reveal that these contact points can be produced in a reputable and easily reproducible way,” claims Walter Weber. “The technological systems you require to do this are currently being used in the chip industry today. This is not just a laboratory experiment, yet a process that might be utilized relatively quickly in the chip industry.”
The manufacturing process’s decisive benefit is that high-grade contacts can be created despite the silicon-germanium composition. “We are persuaded that today abrupt, robust and dependable metal-semiconductor contacts are extremely interesting for a range of new nanoelectronic, optoelectronic and quantum devices,” states Walter Weber.
The research was published in Small.
Read the original article on PHYS.
Read more: Investigators Look Into Combating Tumours With Magnetic Bacteria.