A dangerous superbug responsible for more than a million people worldwide each year might have a natural foe living just beneath your nose. Literally so—it plays a major role in your skin microbiome and appears to help fend off staph infections.
Malassezia sympodialis: A Common Skin Yeast with Hidden Power Against Staph Infections
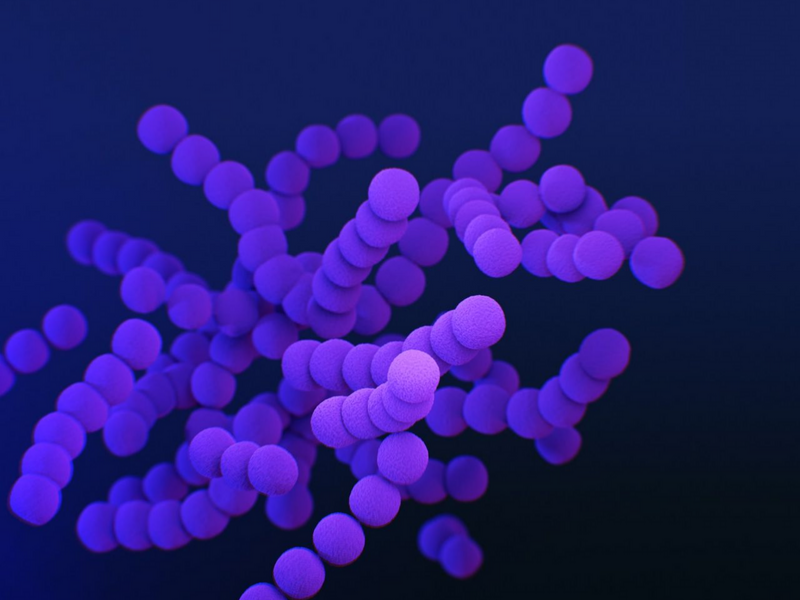
Malassezia sympodialis, a common skin microbe, produces a fatty acid that prevents staph infections and breaks down oils and fats.
Researchers at the University of Oregon led laboratory experiments that showed M. sympodialis suppresses Staphylococcus aureus by releasing acidic byproducts.
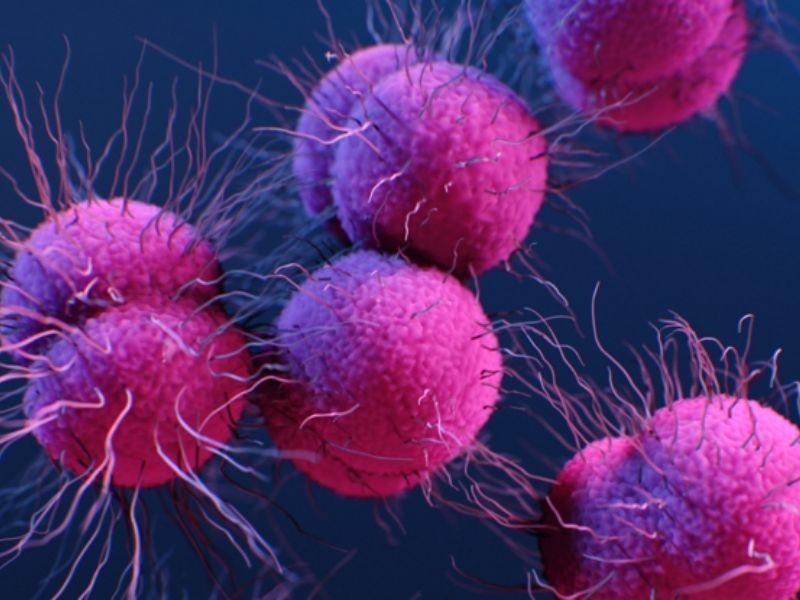
Scientists believe the yeast-generated acid, commonly found on healthy skin, helps prevent S. aureus from taking over the microbiome. S. aureus is usually on the skin but can cause serious infections if it spreads or enters the bloodstream.
In the U.S., skin and soft tissue infections caused by S. aureus account for roughly 500,000 hospitalizations each year. Alarmingly, the bacterium can develop resistance to every class of antibiotics currently available.
This makes the search for new treatment options urgent—and underscores the importance of studying our own skin’s natural defenses. Lead author Caitlin Kowalski says their study is exciting because it highlights a familiar but previously overlooked molecule.

10-HP: A Skin-Activated Antimicrobial Compound Hidden in Plain Sight
10-HP was likely overlooked because it only works in acidic environments like skin, not in typical lab conditions. As a result, researchers overlooked its potential in previous studies.
By examining skin biopsies from healthy donors, Kowalski and her team discovered that the acid is produced by the naturally occurring Malassezia yeast living on the skin.
“It was like searching for a needle in a haystack—but with invisible molecules,” says Matthew Barber, a biologist and adviser to lead researcher Caitlin Kowalski.
In the lab, Barber, Kowalski, and their team tested how M. sympodialis yeast affected different strains of Staphylococcus aureus. After just two hours of exposure, most strains showed over a 100-fold drop in viability.
However, over time, S. aureus began developing resistance to the yeast’s 10-HP compound—using mechanisms similar to how it adapts to clinical antibiotics.
Researchers found that less harmful Staphylococcus species had adapted to coexist with M. sympodialis, hinting at a long-standing microbial balance. Given how common Malassezia is, researchers believe we’re just beginning to understand its role in microbial balance and defense.
Kowalski now plans to explore the genetic pathways behind antibiotic-resistant staph infections to better grasp how these bacteria rapidly evolve to evade various treatments.
“We still have a great deal to learn about these microorganisms,” Barber adds, “and about how we might develop new strategies to treat or prevent the infections they cause.”
Read the original article on: Sciencealert
Read more: Innovative Drug Delivery System Stores Doses as Crystals Beneath the Skin