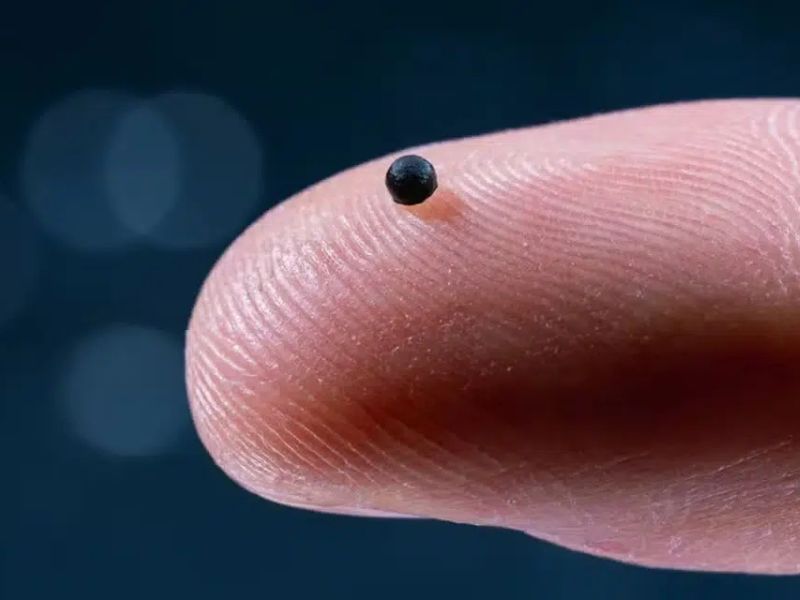
An ETH Zurich grain-sized microrobot has demonstrated in preclinical tests the ability to navigate the bloodstream, deliver drugs precisely, and dissolve, showing promise for stroke and localized cancer treatments.
The system consists of a miniature capsule that holds the drug along with iron oxide nanoparticles. Iron oxide nanoparticles let operators steer the microrobot via magnetic fields and X-ray imaging, then release the drug by heating the capsule with high-frequency pulses.The capsule’s materials then either dissolve or are cleared from the body.
Unprecedented Precision in Targeted Intravascular Delivery
According to the researchers, the experiments showed that over 95% of the drug releases in animal models took place precisely at the intended site—an exceptional level of accuracy for targeted delivery within blood vessels. The demonstrations covered catheter insertion, movement both with and against blood flow, and precise navigation along vessel walls.
- Magnetic control: External magnetic fields enable steering and drug release.
- Real-time imaging: X-ray guidance provides accurate tracking and positioning.
- Biocompatible design: Materials are evaluated in large animals and engineered to break down safely.
Although results in pigs and sheep are promising, the technology is still preclinical; safety, immune response, and human-specific imaging and navigation must be verified before clinical use. The authors note that regulatory evaluations and clinical trials will be required to determine safety and effectiveness in humans.
Reducing Side Effects and Increasing Precision
Delivering many medications systemically can cause toxicity and unwanted side effects. Approaches that deposit small doses directly at the affected site can improve effectiveness while minimizing harm to healthy tissue. Potential early uses include targeted clot breakdown in ischemic stroke, localized therapy for brain tumors, and intricate endovascular procedures requiring high precision.
The ETH Zurich microrobot, featuring magnetic steering, controlled drug release, and post-delivery dissolution, is nearing clinical trial readiness, though regulatory, safety, and imaging challenges remain. If development continues as anticipated, the method could reshape certain invasive treatments in the coming decades.
Read the original article on: Fatos Desconhecidos
Read more: An injectable Weight-loss Drug can Reduce Leftover Post-Surgery Pounds