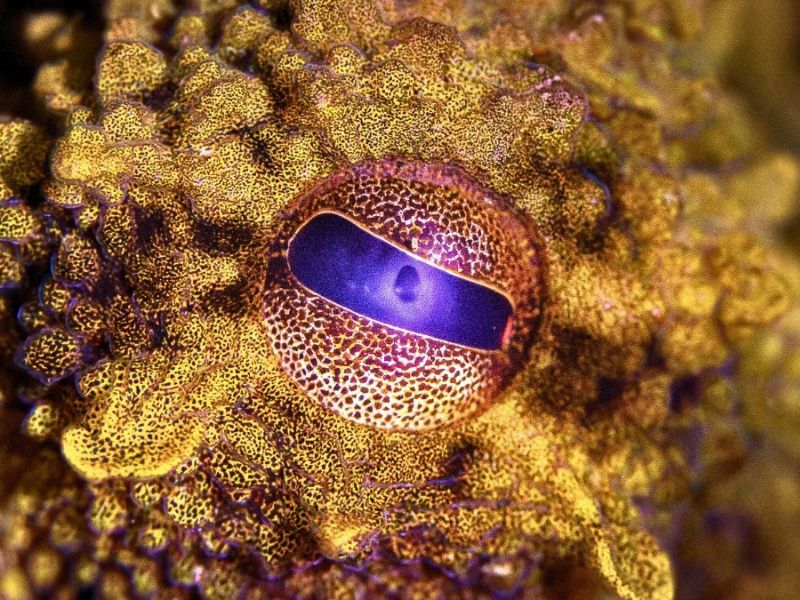
Octopuses and other cephalopods are renowned for their remarkable camouflage abilities, using color-shifting skin to blend seamlessly into their surroundings. Now, scientists have made significant progress toward replicating this natural talent.
UCSD Engineers Bacteria to Mass-Produce Cephalopod Pigment
A UC San Diego–led research team has succeeded in mass-producing xanthommatin — a key pigment found in the vibrant, shifting skin of cephalopods. Previously, this pigment was nearly impossible to harvest from animals or synthesize in a laboratory.
Rather than creating the pigment directly, the researchers engineered bacteria to produce it. These modified microbes generated xanthommatin with exceptional efficiency — up to 1,000 times more than any prior attempts.
Easier access to xanthommatin could advance research into cephalopod camouflage, offering deeper insight into this natural marvel and revealing strategies we might use to replicate it.
Microbial Manufacturing Could Transform Industrial Production
Beyond the pursuit of octopus-like abilities, the study also highlights the potential of microbial manufacturing. If bacteria can be similarly engineered to create other compounds, it could revolutionize industrial production methods.
“We’ve developed a new approach that allows us to rapidly produce a material—in this case, xanthommatin—within bacteria for the first time,” says senior author Bradley Moore, a marine chemist at Scripps Oceanography and the University of California San Diego.
“This natural pigment is what enables octopuses and squids to camouflage—a truly remarkable ability—and our success in scaling up its production is only the beginning,” Moore adds.
Growth-Coupled Biosynthesis Boosts Pigment Production
To coax higher yields from otherwise uncooperative bacteria, the researchers developed a new technique called “growth-coupled biosynthesis.” This method motivates bacteria to produce large amounts of xanthommatin by linking their survival directly to the pigment’s creation.
“We needed a completely new strategy to tackle this challenge,” explains lead author Leah Bushin, who conducted the work in the Moore Lab at Scripps Oceanography.
“Basically, we figured out a way to trick the bacteria into making more of the compound we wanted,” she adds.
Bacteria are inherently efficient, preferring not to expend energy on substances that don’t contribute to their immediate survival.
To solve this, Bushin and her team gave the bacteria an offer they couldn’t refuse. They genetically modified “weakened” cells that could only survive if they continuously produced two molecules: xanthommatin and formic acid.
Formic acid acted as a fuel source, and because each pigment molecule produced was paired with one molecule of formic acid, the bacteria could keep growing—but only if they kept making pigment. This clever feedback loop drove sustained, high-level xanthommatin production.
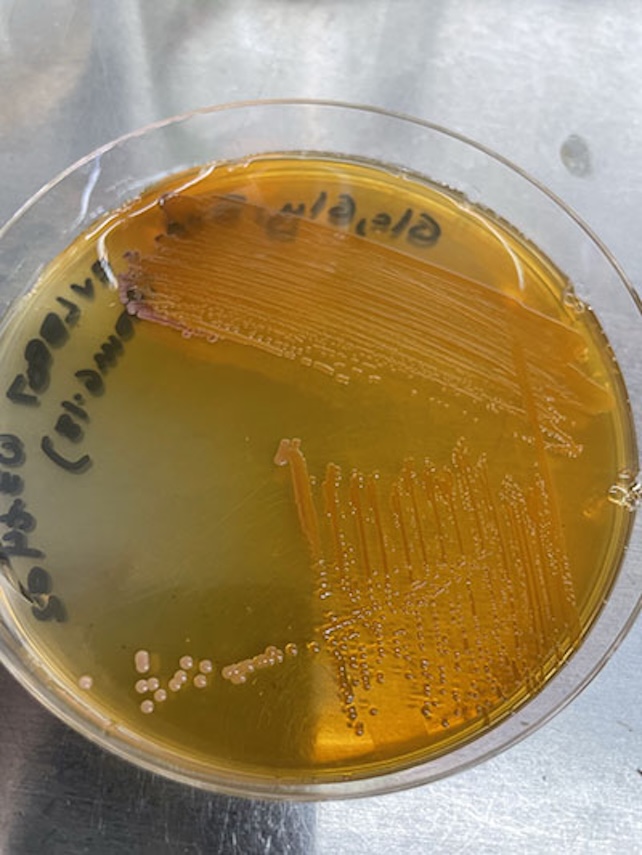
“We designed the system so that the pathway producing the desired compound became essential for survival,” Bushin explains. “If the bacteria stop making xanthommatin, they simply can’t grow.”
New Method Yields Pigment at Record Levels
The method produced up to 3 grams of pigment per liter of culture medium—an enormous improvement over the roughly 5 milligrams per liter achieved with previous techniques, according to the researchers.
Once the setup was established, results began to appear quickly.
“It was one of my best days in the lab,” Bushin recalls. “I set up the experiment and left it overnight. When I came in the next morning and saw that it worked—and that the bacteria were producing so much pigment—I was ecstatic. Moments like that are why I love doing science.”
Beyond the feedback loop that powered their approach, the team further refined their bacterial strains using adaptive laboratory evolution and bioinformatics tools. These enhancements boosted efficiency and enabled the microbes to generate pigment from a single nutrient source, such as glucose.
The findings suggest remarkable promise for this approach, says co-author Adam Feist, a bioengineer at UC San Diego.
“This project offers a look into a future where biology drives the sustainable creation of valuable materials and compounds through advanced automation, data integration, and computational design,” Feist explains.
“Our work demonstrates how collaboration among engineers, biologists, and chemists—combined with cutting-edge strain-engineering methods—can rapidly accelerate innovation in biomanufacturing and enable the development of new products in a fraction of the usual time.”
Read the original article on: Sciencealert
Read more: Magnetic Gel May Offer a more Efficient Way To Eliminate Kidney Stones