Image Credits: Depositphotos
A device resembling a glue gun that prints biodegradable bone grafts directly into fractures has the potential to transform orthopedic surgery by providing customized implants that promote faster healing and reduce infection risk.
Fractures and bone loss from cancer surgeries often leave large defects, typically treated with bone grafts or metal implants. Researchers have explored many approaches to bone repair over the years, including grafts made from eggshells and piezoelectric scaffolds.
Revolutionizing Personalized Bone Repair with 3D-Printed Grafts
In a recent study, researchers from Korea and the U.S. created and tested a modified glue gun capable of 3D printing bone grafts directly onto fractures and bone defects during surgery.
“Our technology introduces a novel approach by enabling in situ printing, which allows scaffolds to be fabricated and applied in real time at the surgical site,” said Jung Seung Lee, co-corresponding author and associate professor of biomedical engineering at Sungkyunkwan University (SKKU), South Korea. “This method ensures precise anatomical fit, even for complex or irregular defects, without requiring pre-surgical steps like imaging, modeling, or trimming.”
The researchers developed a glue gun-inspired device designed for low-temperature 3D printing, making it safe for application on living tissues. The device uses sticks made from a blend of polycaprolactone (PCL)—a biodegradable polymer—and hydroxyapatite (HA), a bone-derived mineral. This composite can be melted and extruded directly into bone defects without the need for toxic solvents. By adjusting the molecular weight of PCL and the HA content, they were able to control the material’s strength, flexibility, and degradation rate. The team evaluated properties such as compressive and bending strength, bone adhesion, and degradation in simulated body conditions. They also successfully incorporated antibiotics like vancomycin and gentamicin into the material.
Preclinical Testing and Real-Time Surgical Control of 3D-Printed Bone Grafts
Researchers initially tested mouse pre-osteoblasts (precursors to bone-forming cells) and human bone marrow stem cells to assess toxicity and support for bone cell growth. Following this, they tested the composite in a rabbit model with a femoral bone defect too large to heal on its own, comparing the performance of their 3D-printed material to commercial bone cement over a 12-week period.
“Since the device is compact and manually operated, surgeons can control the printing direction, angle, and depth in real time during surgery,” said Lee. “We also demonstrated that surgeons can complete the entire process within minutes, providing a major advantage by reducing operative time and improving efficiency in real surgical settings.”
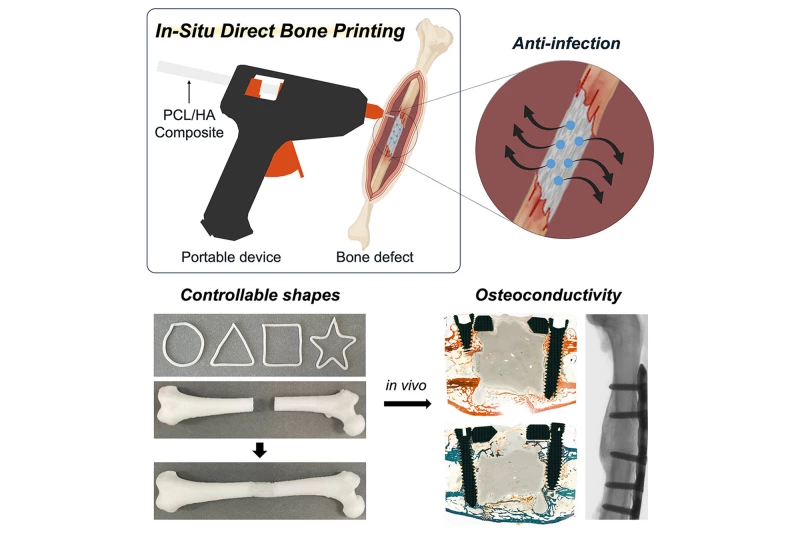
Jeon, In Yeop et al. (2025)
Enhanced Scaffold Performance and Antibacterial Properties Through Hydroxyapatite Integration
The researchers discovered that adding hydroxyapatite (HA) to the composite improved its strength and elasticity, while also slowing its degradation—allowing the scaffold to remain in place long enough to support new bone growth. Higher molecular weight PCL enhanced the material’s adhesion to bone, though excessive HA slightly reduced this effect. Lab tests showed that scaffolds infused with antibiotics effectively inhibited bacterial growth, with gentamicin showing particularly strong results. No toxicity was observed. HA also promoted bone cell attachment, growth, and the transformation of precursor cells into mature bone cells.
“This targeted delivery method offers clear clinical benefits over systemic antibiotic use by minimizing side effects and the risk of antibiotic resistance, while still providing strong protection against post-surgical infections,” said Lee.
Animal Trials Show Superior Bone Regeneration with 3D-Printed Scaffolds
In animal trials, the 3D-printed scaffolds outperformed conventional bone cement in supporting new bone formation. Micro-CT scans taken 12 weeks after surgery revealed that the printed material led to stronger, more natural bone development, without any tissue damage or abnormal inflammation. However, researchers observed that although bone healing improved significantly compared to bone cement, the defects had not fully filled by the end of the study period.
“The scaffold was designed to biologically integrate with surrounding bone tissue and to gradually break down as new bone replaces it over time,” said Lee. “Our findings showed that the printed scaffolds led to better results in key structural indicators like bone surface area, cortical thickness, and polar moment of inertia, all pointing to more effective bone regeneration and integration.”
Key Bone Strength Metrics and the Future of Personalized Orthopedic Grafting
Cortical thickness refers to the dense outer layer of bone and is a key marker of bone strength and density. The polar moment of inertia measures a bone’s resistance to twisting forces, reflecting its structural stability.
This proof-of-concept bone grafting technique could revolutionize personalized orthopedics. Surgeons might bring this portable device into the operating room to print customized bone implants directly onto irregular defects. The built-in antibiotic delivery system could help reduce post-surgical infections, a leading cause of implant failure. This method would be faster and more cost-effective, eliminating the need for expensive, time-consuming implant manufacturing. Additionally, the device can adapt to use different biodegradable materials and incorporate various drugs to address diverse bone injuries and patient needs. However, researchers need to conduct further work.
“Clinical use will depend on establishing standardized manufacturing, validated sterilization methods, and preclinical testing in large animal models to meet regulatory standards,” said Lee. “If researchers overcome these challenges, we envision this technique becoming a practical, immediate solution for bone repair directly in the operating room.”
Read the original article on: New Atlas