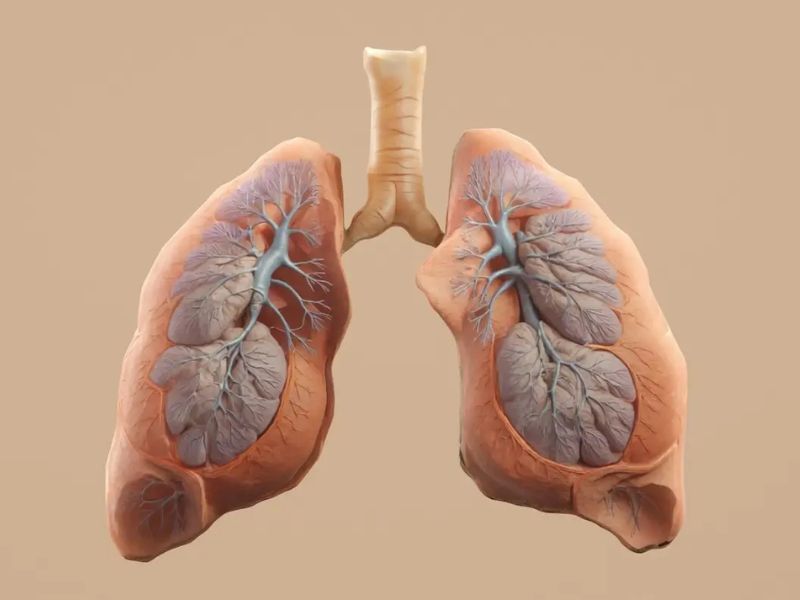
A genetically engineered pig lung functioned for 216 hours inside a brain-dead human, marking the first reported attempt at a cross-species lung transplant, according to Nature Medicine.
The technique, known as xenotransplantation, aims to ease the chronic shortage of donor organs. Pig organs are similar in size to human ones, but their proteins often trigger severe immune rejection. Over time, scientists have pinpointed the genes behind these problematic proteins and used gene-editing tools to make pig organs more compatible with the human body.
China Pushes Boundaries With First Pig Lung Transplant
Researchers have already transplanted modified pig hearts, livers, and kidneys into people under experimental protocols, showing encouraging progress. Now, a team at the First Affiliated Hospital of Guangzhou Medical University in China has added lungs to that list—though with important limitations.
The lung transplant lasted more than nine days but eventually caused damaging inflammation, despite an intensive regimen of immune-suppressing drugs.
Still, the researchers note that their results “pave the way for further innovations in the field.”
Every day, about 13 people die while waiting for an organ transplant. The problem is stark: there simply aren’t enough donor organs.
For a transplant to succeed, the donor organ must be closely matched to the recipient’s blood type and immune markers, which makes the wait agonizingly long. As of late September 2024, nearly 90,000 patients were on the kidney transplant list, while more than 3,000 awaited a new heart.
Pig organs offer a possible alternative—but in their natural state, they’re unsafe for humans.
Viruses and Rejection
One issue is that pig DNA carries porcine endogenous retroviruses (PERVs). These viruses don’t harm pigs but can infect humans. Another is immune rejection: every organ is covered in protein markers, like a biological fingerprint. If the body doesn’t recognize that fingerprint, the immune system mounts an aggressive defense. Killer T cells, B cells, and inflammatory molecules known as cytokines can overwhelm and destroy the transplant.
The solution is to make pig organs more human-like so they evade immune detection.
Over the years, researchers have identified the pig genes that encode these problematic proteins and used CRISPR-Cas9 to remove them. But that created new challenges: without certain protein signals, the organs looked abnormal to immune cells. To counter this, scientists inserted three human genes that regulate immune responses, essentially camouflaging the organs.
After years of refinement, Chinese researchers developed a genetically altered Bama Xiang pig—a small breed native to southern China—with six modified genes designed to make its organs more compatible with humans.
At least in theory
First Pig Lung Transplant Attempt in Humans
In a recent trial, scientists transplanted the left lung of a genetically modified Bama Xiang pig into a brain-dead 39-year-old man, with his family’s consent. The organ tested virus-free, and the surgery largely followed standard lung transplant procedures, though some pig structures had to be trimmed for fit.
Lungs break more easily than other organs, and restoring blood flow can severely damage them. Yet within a day the transplanted lung stabilized and functioned normally. By day two, however, it showed signs of acute rejection, with swelling, immune cell activity, and later spikes in antibodies. By day nine, the lung had partially healed and was exchanging oxygen, but the trial ended at the family’s request.
Researchers detected no pig viruses during the study, and patients developed no infections despite immune suppression. Lungs face unique hurdles—high blood pressure, pathogen exposure, and rejection-prone proteins. In this trial, the immune response was faster and stronger than in baboons, highlighting the need for better drugs or more genetic edits.
The team now plans to test existing transplant drugs—and potentially add blood thinners or anti-inflammatories—to better control lung-specific immune reactions in future trials.
Read the original article on: Singularity Hub